Adolescents Between 12 -15 Years Are Earmarked For Pfizer Vaccine On FDA’s Approval
Nikki Attkisson | Last Updated : May 5, 2021Pfizer’s Covid-19 vaccine is set to be authorized by the U.S. Food and Drug Administration as early as next week. Thereby setting the younger adults 12 years and older eligible for vaccination. This was corroborated by a federal official associated with the vaccination process. The move of setting up shots comes ahead of the resumption of school early next year.
Adolescents Between 12 -15 Years Are Earmarked For Pfizer Vaccine On FDA’s Approval
Studies have confirmed that the Pfizer vaccine approved for people aged 16 years and above also works for the younger lot. The announcement was hailed a month post the authorization of vaccinating individuals 16 years and above in the age bracket.
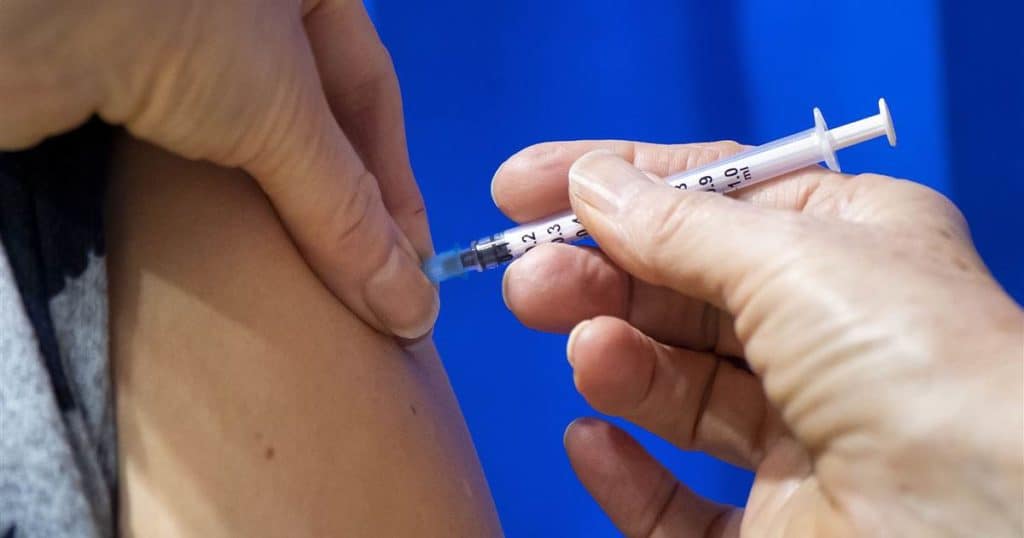
Several reports are doing the rounds as confirmed by a federal official who chose to stay anonymous in regards to the preview of action by the FDA. The two-dose vaccine by Pfizer is set to be upgraded for emergency usage, and plans of expansion are up as early as next week or could be sooner.
The official also mentioned the timeline and stated how FDA is up for approval of the Pfizer vaccine amongst the younger children close to this fall.
A federal vaccine advisory committee is scheduled for a meeting to discuss the recommendations cited for the 12- to 15-year-old recipients of the vaccine shot. This is slated to get a breather upon the adoption of the recommendations by the Centers for Disease Control and Prevention. The same is supposed to be completed within a few days.
The authorization timings were first corroborated by “The New York Times.” The preliminary set of results post the data compilation of the vaccine was amongst 2260 volunteers in the U.S., all of whom were children aged between 12 to 15.
The reports were released in late March, which confirmed there were zero cases of Covid-19 amongst the fully vaccinated teens. This was in comparison to the eighteen adolescents who received dummy shots.
The kids reported showcasing similar side effects that are most likely to occur amongst younger adults. This included pain, chills, fever, and fatigue, mainly which occurred post receiving the second shot of the vaccine. Some studies are ongoing to track the participants over two years to increase the efficacy of the vaccine as well as provide long-term protection.
The lowering of the age limit for its vaccine has even led Moderna to round up a study to test the vaccine’s efficacy and the participants include 12- to 17-year-olds. The results are awaited in the second half of the year.
The FDA found a promising sign in its findings that led to the two vaccine major companies getting the approval for the commencement of the studies amongst children. The age bracket as categorized is 11 and below, going down to tots aged six months.
Pfizer has achieved a milestone in administering 131 million vaccine doses across the U.S, thus leading to a dramatic decline in the vaccine rush in recent times.
The risk of serious side effects emanating from Covid-19 is much lower amongst younger individuals. However, the current scenario showcases the new infection load is mostly high amongst the younger people.
This is attributed to two different reasons. One, since the adults have received either one or two doses of their vaccine shots as well as the resumption of high-risk activities. This includes indoor dining, contact sports, etc.
There is still hope in the offing, as cited by the federal officials, once the vaccinations are extended to accommodate teens. This will further see a better acceleration in reducing the caseload. Thus, allowing schools to open up this fall with bare minimum disruptions.
The Pfizer vaccine requirements have shot up, with the U.S. ordering approximately 300 million doses within July to protect its 150 million people.
With over 15 years as a practicing journalist, Nikki Attkisson found herself at Powdersville Post now after working at several other publications. She is an award-winning journalist with an entrepreneurial spirit and worked as a journalist covering technology, innovation, environmental issues, politics, health etc. Nikki Attkisson has also worked on product development, content strategy, and editorial management for numerous media companies. She began her career at local news stations and worked as a reporter in national newspapers.
