After The Adults, It Is Time For Kids Now To Get Their Shots
Nikki Attkisson | Last Updated : February 3, 2022Vaccines have acted as a savior for many so far, and health experts all over are urging people to get their shots. Immunization of adults had started a long time back, but the emergence of the new Omicron, and the few reported severities in children, have left many wondering as to when the youngest generations would get their lifesaving doses of vaccine, more so, when most of the schools around, are planning to reopen.
After The Adults, It Is Time For Kids Now To Get Their Shots
The US Food and Drug Administration had given Pfizer-BioNTech the approval of providing vaccines to children till the age of 5 but not below that. However, it is aiming to extend its services to infants as young as 6 months of age soon.
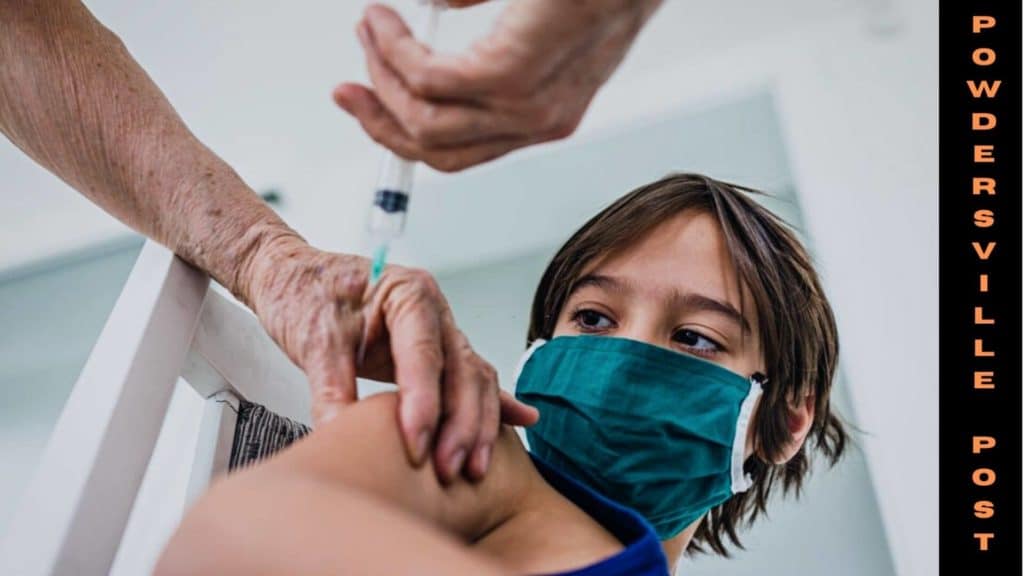
The processes have already started with submissions of documents to the FDA, but the FDA has to decide on how many doses should a child of that age group require? Trials on infants have shown that two doses are enough to provide protection to infants but not so to preschool goers.
They announced that one-tenth of the dose that is administered to adults is used for vaccinating children below the age of 5, but the results are quite confusing as they have seen very little response against the virus, in children of that age group.
The experts hypothesize that the doses must be too low for children aged 2 to 4 years. And hence, they have agreed on the administration of two doses with a three-week gap followed by a third at least after two months.
Now it is the turn of the FDA to approve the decision and give the final nod to the proposal. It plans to set up a panel of experts to review the data provided by Pfizer and finally decide on the number of doses.
Pfizer CEO, Albert Bourla, feels that the FDA will ultimately agree on the three shots, but till it gives its final verdict, it should allow parents to proceed with the vaccinations.
As the number of infection cases in children under the age of 5 is surging, the FDA had asked Pfizer to submit its papers earlier as stated by one of the FDA’s spokeswomen, Stephanie Caccamo. But, the decision does not lie only in the hands of the FDA as the Centers for Disease Control and Prevention also has to approve of the plans.
As Dr. Sean O’Leary of the University of Colorado said, the number of increased hospitalizations and deaths amongst the youngest Americans is really unfortunate, and hence, the FDA’s approval for vaccination of those children should aid in minimizing the numbers and saving the lives of many.
There are almost 19 million children below the age of 5 who are still not eligible to get their shots while the administration is trying hard to provide them with the facilities.
Vaccines are critical at this point in time as most of the states are planning to resume in-person classes, and also the daycare centers need to open up so that the parents are free to work again. But, not many children have received their shots yet.
As reported by the American Academy of Pediatrics, till last week, only 20 % of the children between the ages of 5 and 11 have got immunized fully and only half of the young generation of 12 to 17 years has been inoculated.
And what is more concerning is that only a few parents are willing to give their children the advantage of vaccines while most of them are still reluctant in doing so.
Although data proves that children are much less likely to contract severe illness from this Omicron, increasing numbers of fatalities at this point of the pandemic are pointing towards the need for vaccination approval. Many are awaiting the decision which FDA should be provided with very soon.
With over 15 years as a practicing journalist, Nikki Attkisson found herself at Powdersville Post now after working at several other publications. She is an award-winning journalist with an entrepreneurial spirit and worked as a journalist covering technology, innovation, environmental issues, politics, health etc. Nikki Attkisson has also worked on product development, content strategy, and editorial management for numerous media companies. She began her career at local news stations and worked as a reporter in national newspapers.
